|
Pembrolizumab or placebo were continued until developmental of progressive disease, up to 35 total cycles of treatment or other discontinuation criteria were met. Chemotherapy was administered every 3 weeks for up to 4 cycles. Chemotherapy consisted of carboplatin plus a taxane (paclitaxel or nab-paclitaxel). KEYNOTE-407 was a randomized, double-blind, phase III trial comparing pembrolizumab plus chemotherapy (n=278) versus placebo plus chemotherapy (n=281). KEYNOTE-407 demonstrated an improvement in both PFS and OS for the addition of pembrolizumab to chemotherapy ( 13). 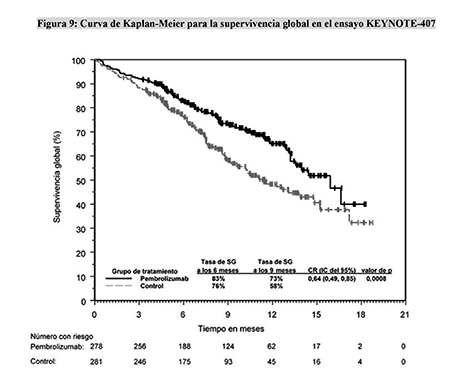
KEYNOTE-407, which is the focus of this editorial commentary, evaluated the addition of pembrolizumab to carboplatin plus taxane. IMpower131 demonstrated a PFS benefit for the addition of atezolizumab, but no OS benefit ( 11, 12). IMpower131 evaluated the addition of the programmed death ligand-1 (PD-L1) inhibitor atezolizumab to carboplatin plus nab-paclitaxel. The cytotoxic T-lymphocyte antigen-4 (CTLA-4) inhibitor ipilimumab combined with carboplatin and paclitaxel did not result in improved PFS or OS when compared to chemotherapy alone ( 10). Similarly, three phase III trials evaluated the addition of an immune checkpoint inhibitor to platinum-based doublets in advanced squamous NSCLC ( 10- 13). The PD-1 inhibitor pembrolizumab added to first-line platinum-based chemotherapy demonstrated superior objective response rate (ORR), progression-free survival (PFS) and OS in non-squamous NSCLC ( 9). Immune checkpoint inhibitors were then evaluated as initial treatment in combination with platinum-based chemotherapy. 
As a result, PD-1 axis inhibitors became the preferred second-line therapy after development of progressive disease on a platinum-based doublet. Subsequently, four randomized phase III trials demonstrated significant improvements in survival when compared to docetaxel for patients receiving monotherapy with a programmed death-1 (PD-1) axis inhibitor ( 5- 8). Standard of care for patients with squamous NSCLC who developed progressive disease on platinum-based doublets was either (I) docetaxel with or without ramucirumab or (II) gemcitabine ( 3, 4). Median overall survival (OS) with these regimens was 10–12 months and 5-year OS was estimated at 2% ( 1, 2). Patients with stage IV squamous non-small cell lung cancer (NSCLC) were historically treated with first-line platinum-based therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
